|
"Reactions of Main Group Elements with Water". "Reactions of the Group 2 Elements with Water". ^ "Reactions of Group 2 Elements with Water".Michael Pilgaard's Table of the Elements. REACTPOOL: A new model for accidental releases of water-reactive chemicals (PDF). Archived from the original (PDF) on 26 February 2018. "MODELLING RELEASES OF WATER REACTIVE CHEMICALS" (PDF). Archived from the original on 3 March 2018. Interactive Learning Paradigms Incorporated. ^ "The MSDS HyperGlossary: Metal Reactive".The reaction of fluorine with water is especially hazardous, as an addition of fluorine gas to cold water will produce hydrofluoric acid, oxygen gas, and ozone. Out of the four stable halogens, only fluorine and chlorine have reduction potentials higher than that of oxygen, allowing them to form hydrofluoric acid and hydrochloric acid directly through reaction with water. Halogens are so named due to their potential to form salts, and form many simple strong acids with hydrogen. Hydrogen is always produced when a metal reacts with cold water or steam. If metals react with steam, oxides are formed.If metals react with cold water, hydroxides are produced. 
Slow reaction with cold water, vigorous with hot water Reactivity series of metals Order of reactivity Notably, radium hydroxide is the most soluble out of all alkaline earth hydroxide species. 
Radium reacts similarly to the rest of the alkaline earth metals (other than magnesium), forming radium hydroxide and hydrogen gas. The hydroxides of calcium, strontium and barium are only slightly water-soluble but produce sufficient hydroxide ions to make the environment basic, giving a general equation of: However, if a metal reacts with steam, like magnesium, metal oxide is produced as a result of metal hydroxides splitting upon heating. Mg(s) + 2H 2O(l) ⟶ Mg(OH) 2(s) + H 2(g) Ī metal reacting with cold water will produce a metal hydroxide and hydrogen gas. The reaction is short-lived because the magnesium hydroxide layer formed on the magnesium is almost insoluble in water and prevents further reaction. Magnesium has a mild reaction with cold water. Magnesium shows insignificant reaction with water, but burns vigorously with steam or water vapor to produce white magnesium oxide and hydrogen gas: Mg(s) + H 2O(g) ⟶ MgO(s) + H 2(g) Additionally, beryllium has a resistant outer oxide layer that lowers its reactivity at lower temperatures. Beryllium (Be) is the only alkaline earth metal that does not react with water or steam, even if the metal is heated red hot. The alkaline earth metals (Be, Mg, Ca, Sr, Ba, and Ra) are the second most reactive metals in the periodic table, and, like the Group 1 metals, have increasing reactivity with increasing numbers of energy levels. 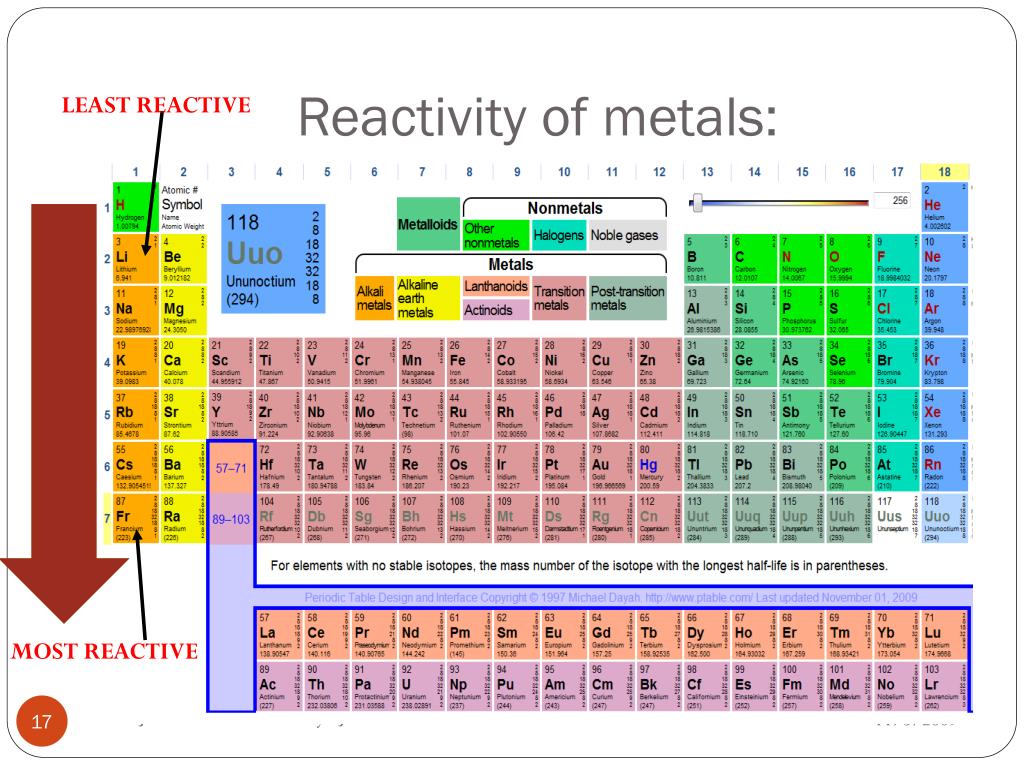
Main article: Alkaline earth metals Group 2: Alkaline earth metals In an NFPA 704 fire diamond's white square, they are denoted as "W̶".Īll chemicals that react vigorously with water or liberate toxic gas when in contact with water are recognized for their hazardous nature in the 'Approved Supply List,' or the list of substances covered by the international legislation on major hazards many of which are commonly used in manufacturing processes. Such substances are classified as R2 under the UN classification system and as Hazard 4.3 by the United States Department of Transportation. The use of acid-resistant gloves and face shield are required and should be handled in fume hoods. Some water-reactive substances are also pyrophoric, like organometallics and sulphuric acid, and should be kept away from moisture. Notable examples include alkali metals, lithium through caesium, and alkaline earth metals, magnesium through barium. Water-reactive substances are those that spontaneously undergo a chemical reaction with water, as they are highly reducing in nature. Substance that readily reacts with water in a potentially hazardous way
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
