|
F ormula Shape Bond Angle ( Theoretical ) AX 2 Linear 180 AX 3 Trigonal Planar 120 AX 4 Tetrahedral 109.5 AX 5 Trigonal Bipyrimidal 120, 90 AX 6 Octahedral 90 AX 3 N Trigonal Pyramidal 109.5 AX 2 N Bent 120 AX 2 N 2 Bent 109.5įrom the above table, it can be observed that an AX 3 N arrangement corresponds to a Trigonal Pyramidal Molecular geometry. Therefore, that would give us an AX 3 N arrangement for Arsenic Trifluoride.įrom the A-X-N table below, we can determine the molecular geometry for AsF 3. In this case, N = 1 as there is one lone pair attached to the Arsenic atom. ‘N’ represents the number of lone pairs attached to the central atom. In this case, three Fluorine atoms are bonded to the central Arsenic atom. ‘X’ represents the number of atoms bonded to the central atom. ‘A’ here represents the central Arsenic atom. We can use the A-X-N method to confirm this. This gives it a trigonal pyramidal molecular geometry. The Lewis structure observed tells us that the Arsenic has a steric number of 4 with three covalent bonds and one lone pair attached. Molecular geometry is one of those properties, and we must understand the elemental arrangement from the Lewis structure to proceed. AsF 3 Molecular Geometry and ShapeĪs we discussed earlier, the Lewis structure of a compound helps determine its various properties. This gives AsF 3 bond angles of approximately 96°. 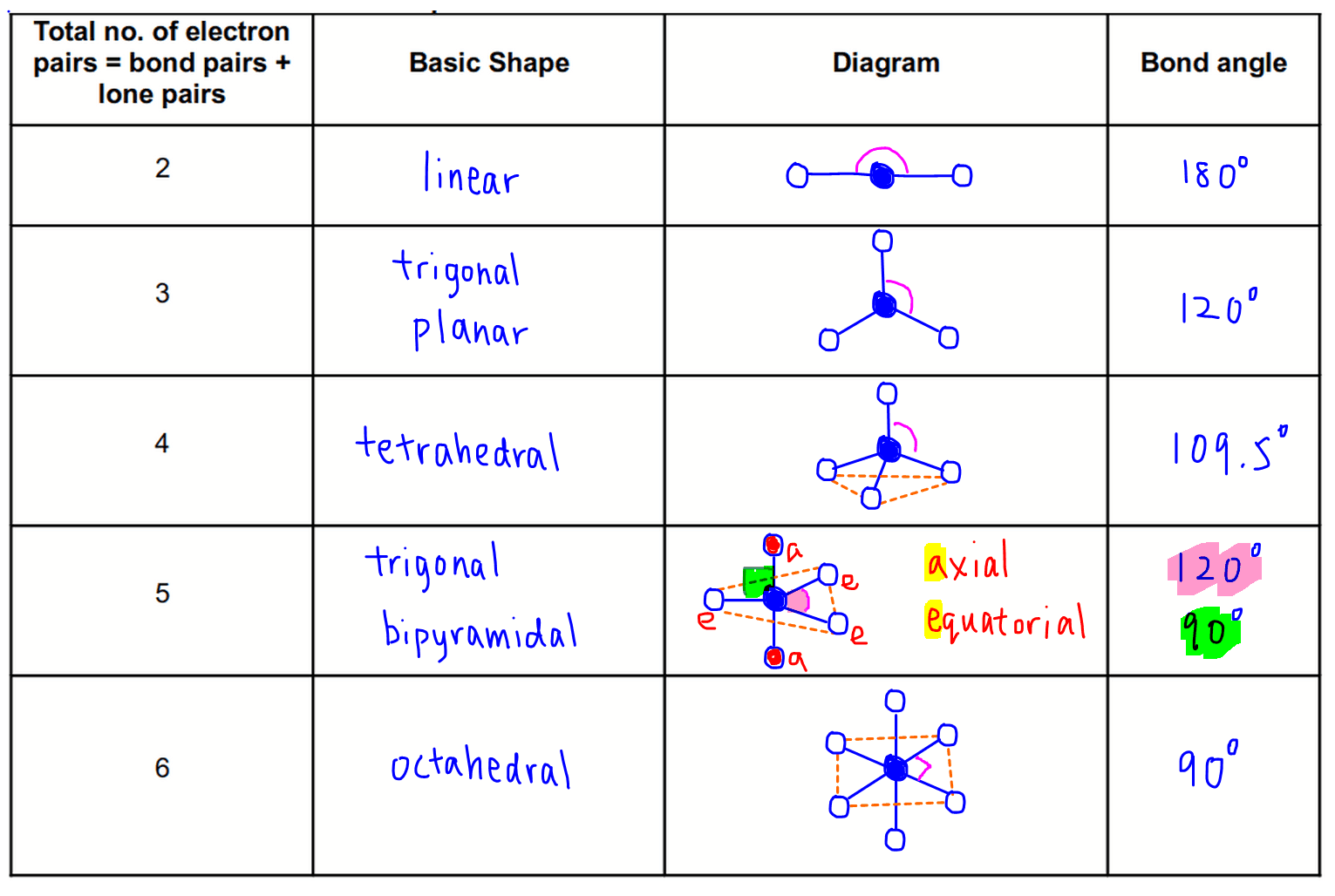
As such, the Fluorine atoms repel each other accordingly. VSEPR theory dictates that like electron pairs repel each other. Therefore, the hybridization of Arsenic in AsF 3 is sp 3. This means that there are four electron domains. In addition to this, there is also a lone pair on the central Arsenic atom. The hybridization of a compound gives information about its energy levels, orbital structure, and the nature of the bonds.ĪsF 3 comprises three covalent bonds between the central Arsenic and Fluorine atoms. The formal charges being 0 for all of the atoms in the AsF 3 molecule tells us that the Lewis structure obtained above is stable. It is determined such that the elemental charge on each atom is closest to zero.įC = Valence Electrons – Non-bonding electrons – (Bonding electrons ÷ 2) We can verify its stability by calculating the formal charges of each constituent atom in the AsF 3 molecule.įormal charges for an element/structure help determine its most stable Lewis Structure state. With this, our Lewis Structure is complete. This will act as a lone pair and will be attached to the central Arsenic atom to fulfill its octet requirements. Valence electrons are used to fulfill the outermost shells of the surrounding Fluorine atom.įinally, this leaves us with two valence electrons. The outermost shell requirements of atoms are addressed in accordance with the octet rule. Covalent bonds between Arsenic and Fluorine are formed by making use of the available valence electrons. Arsenic takes its place as the central atom and facilitates bond formation. Next, we arrange Arsenic and Fluorine atoms in a sort of skeletal arrangement. This has been calculated in the previous section to be 26. 
We already know the number of valence electrons available to us. Lewis structures use the valence electrons contributed by the constituent atoms to show the bond formation and octet fulfillment. Lewis structures are stable schematic representations of the arrangement of atoms in a given molecule. AsF 3 Lewis StructureĪs you already know, the Lewis structure of a molecule or compound helps give insight into various molecular properties. Therefore, the total number of valence electrons in Arsenic Trifluoride is given by:ĥ + 21 = 21 valence electrons. Therefore, the three Fluorine atoms present contribute: 7 x 3 = 21 Valence Electrons. 
It belongs to group 17 of the periodic table and has the electronic configuration 2s 2 2p 5. Therefore, the single Arsenic atom contributes 5 x 1 = 5 valence electrons.įluorine is a halogenic compound. Īrsenic Trifluoride comprises three Fluorine atoms and one Arsenic atom.Īrsenic is in group 5 of the periodic table with the electronic configuration 3d¹⁰4s☤p³. Let us calculate the number of Valence electrons in AsF 3. Using the number of valence electrons available to us, we can determine the Lewis structure for a given compound. This allows the valence electrons to break free from the atom to take part in chemical reactions. These valence electrons reside in the outermost shell of an atom, where the force of attraction from the nucleus is weak. Valence electrons are used in Lewis Structures to represent atomic bonds between atoms and electrons possessed by the individual atoms. AsF3 Molecular Geometry and Shape AsF 3 Valence Electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
